Ministère de l'enregistrement des produits et de l'autorisation du commerce du Bénin
Aux fins de protection de la santé publique

Welcome to MOHBENIN
Welcome to the official website of the Benin Ministry for Products Registration and Trade Authorization Commission (MOHBENIN).
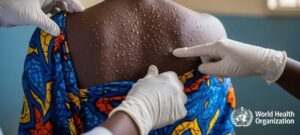
Our mission is to ensure the quality, safety, and efficacy of medicines and health products available in Benin through rigorous registration, inspection, market surveillance, and pharmacovigilance.
Whether you are a pharmaceutical company, healthcare professional, partner institution, or citizen, this platform provides clear information, procedures, and services to support a safer and healthier Benin.
Thank you for visiting us
Director General [MOHBENIN]
800+
Approved Products
60+
Inspection Missions
150+
Certified Facilities






Copyright © 2026 by Sante MOHBenin. All Rights Reserved